
by Ellen Vaders | Apr 24, 2017 | alloy, aluminium, corrosion
Aluminium is known for its corrosion resistance. Untreated aluminium applications like traffic signs and applications at sea are respected examples. Although aluminium is a relatively non noble material it protects itself via a passive oxide layer. Corrosion resistance of Aluminium is sensitive upon certain points in particular, these will be addressed in this blog.
To design aluminium without corrosion take the following aspects into account.
- pH value of the environment
- Nobility of Aluminium
- Crevice corrosion
- Composition of Aluminum
- Black corrosion
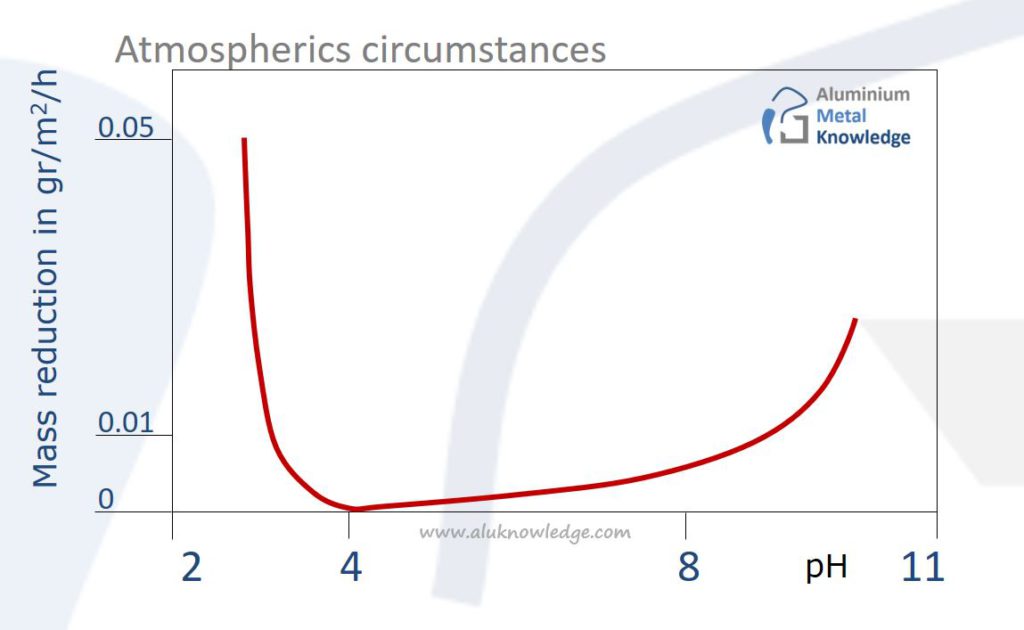
pH value of the environment
Corrosion resistance of Aluminium is influenced strongly by the pH value of the environment
For normal use, like cleaning, use fluids in combinations with Aluminium with a pH neutral value (7) to prevent corrosion.
To prepare Aluminium for surface treatment you can use this to principle to clean the surface with specific sour fluids. The surface needs to be neutralized and rinsed adequately afterwards to prevent corrosion issues!
Nobility of Aluminium
To avoid corrosion problems with Aluminium nobility of the material needs to be taken into account. Aluminium is a relatively non noble material and will therefore sacrifice itself on behave of more noble metals in moister environment. This leads to galvanic corrosion.
This is a serious aspect using Aluminium products, specialy in combination with other metal parts. To prevent this type of corrosion block out fluids, use the right combinations of metals in the same nobility level or isolate the metals from each other.

Crevice corrosion
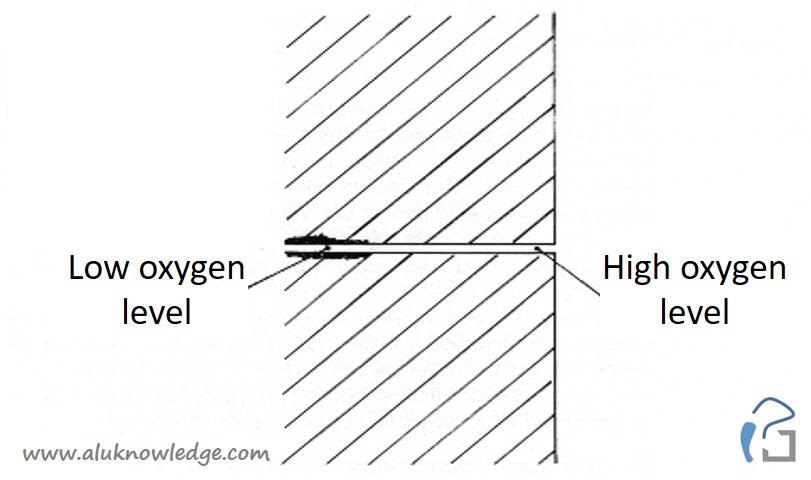
In a moister environment fluid will creep between narrow crevice due to capillary motion. To prevent crevice corrosion you have to avoid small crevices. Crevice corrosion occurs between crevices with a distance in between 0.2 mm to 0.5 mm. The solution for this matter is to increase crevice distance and reduce amount of contact surfaces. For example: a line contact is better than a surface contact.
With acid or salt fluids the potential for crevice corrosion increases.
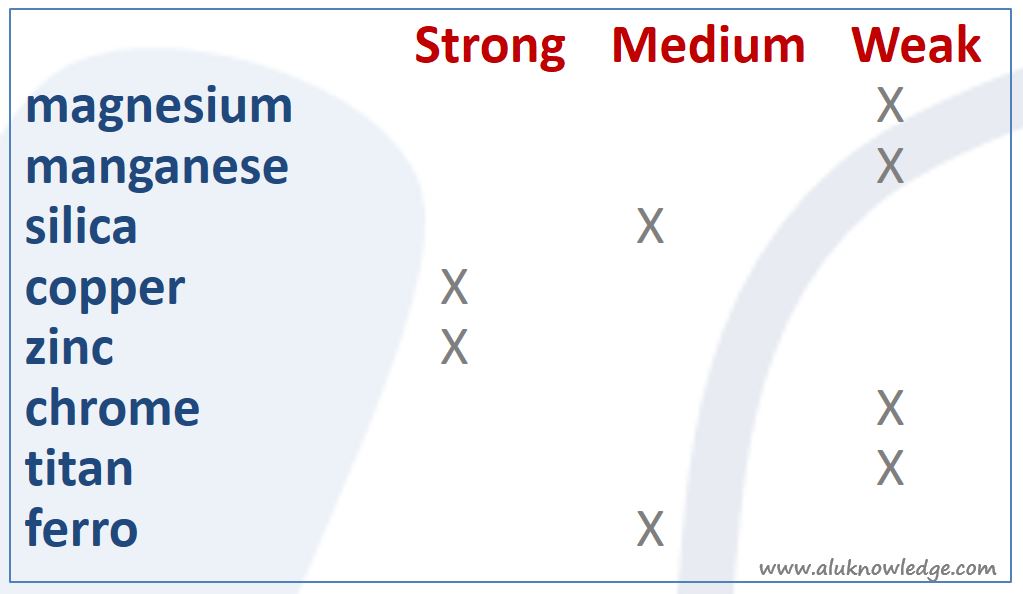
Composition of Aluminum
Last but not least, the composition of the aluminum alloy influences corrosion resistance of Aluminium. If corrosion resistance is low you can surface threat this material in different ways. Take into account it needs more effort than with better corrosion resistance material.
If you take these mechanisms into account your design is robust against most common corrosion issues. In that case you take advantage of the natural corrosion resistance of Aluminium.
What was your biggest insight about corrosion issues so fare?
Let me know in the comment box below. This way, you’re helping me and others to design a world with optimal Aluminium use… Thanks in advance!

by Ellen Vaders | Aug 28, 2015 | alloy, aluminium, corrosion
Op verzoek van een van mijn klanten is dit BLOG in het Engels
On request of one of my customers this new BLOG is in English.
Aluminium is a corrosion resistant material and used frequently, even uncoated, in marine and offshore application with success. For various reasons surface treatment can be required, e.g. visual appearance or specific technical protection.
In all cases corrosion control starts at the construction table, with or without surface treatment.

Still, for coated products, corrosion can occur. Is it related to the use of aluminium or the way we use it?
In most cases problems can be avoided! As mentioned in the BLOG corrosion defects …
there are certain aluminium specific situations, to be aware of while using aluminium. For example, pH of the environment and nobility of the metals used in combination. If you would like to read that BLOG in English please make your request here and I will translate this for you.
This Blog focusses on the aspects to consider in order to prevent corrosion on coated aluminium.
Those are the aspects:
• Design for corrosion
• Material selection
• Pre-treatment
• Coating system dedicated to the situation
The design, to prevent corrosion underneath a coating layer make sure the coating system is closed and has covered the entire part. Crevices, blank spots, or local tiny coating layers can be devastating, corrosion starts from these areas. Also chambers where water may remain will not be of any help for corrosion protection and should be prevented.

Aluminium itself is, depending on the alloy, A or B …, corrosion resistant. Alloy with copper or zinc for instance are more sensitive for corrosion as many of the aluminium magnesium alloys. Coaters have learned from experience that the corrosion index is also a good indicator for coat ability, but it is too complex just to use this “standard rule” as “the only truth” (or law). Knowledge at the coating partner and experience with the selected alloy are of great importance.
Pre-treatment is essential for coating on aluminium. If a company is not familiar with aluminium it will go wrong, with e.g. “delamination” of the coating layer as result. Aluminium can be coated in all kind of colors but doing so, takes quit some investments. A well applied pre-treatment plays a major role to paint adhesion and therefore is important for the overall protection.
Last but not least the coating system. What is the environment for your aluminium product? Is it in the middle of the dessert and should it be painted Ferrari red? Or is your application an aluminium coated trailer with marks on it and in severe dynamics circumstances? Every different situation needs another solution. If the basics are right the issues can be solved.
If all the issues are covered with the right knowledge or partners you will have lifetime well preserved aluminium products.
Please take good care of your aluminium product. Reminder; If possible clean regularly, with pH neutral cleanser! Together we can create a better world with the right use of aluminium!
Would you like to be informed about the possibilities with aluminium?
Subscribe to our mailing list.
Did you face difficulties with your coated aluminium products? Please leave your reaction below.







