
door Ellen Vaders | feb 26, 2018 | aluminium, aluminium advies, aluminium expertise, extrusie, lassen, optimaal ontwerp
What is the length of the Heat affected zone, HAZ?
.
Engineers would like to have rules of thump.
So if we make a welding connection between tubes in the range of 50mm what would the size of the HAZ be? Although I would love to make the world of the engineers a bit easier this question cannot be addressed by a quick answer.
Due to the heat impact the material properties are negatively influenced. The higher the heat input is the more the mechanical properties of the alloys are influenced, for aluminium in the 5000 as well as in the 6000 series. For a tube in the 6000 alloy’s series this means that the mechanical values of a tube in T6 quality are approximately degraded to the T4 value at best (after a recovery period of ~3 months).
How come we can not give a straight answer to the question what length of the area is influenced?
The length of the HAZ is influenced by the heat input of the welding process. So the process type but also all the parameters of the welding process. (current, arc voltage, travel speed, etc.)
.
Two tips for the best result:
1. For MIG welding high current, high travel speed. (snoerend lassen NL)
2. For multilayer welds the interpass temperature of 50° C should be respected, independent of material thickness.
With these tips heat input is relatively low, full penetration in the weld is received, lack of fusion prevented and mechanical strength optimal as possible.
.
You can compare it with preparation of the famous Dutch kroket (Croquette). Welding aluminium is optimal if the heat input is balanced in the right way, for a kroket; shortly hot fried, delivering a nice brown crispy crust is and the meat inside warm but still relatively cold.
.

.
Welding an aluminium tube to a casted bracket the bond-design is somewhat “secured”. If the heat input was too high the weld pool will sag through the weld, if the heat input is too low there is not enough fusion in the weld. Both ways the weld is disqualified.
For specific applications the HAZ can be determined, but if welder or weld supplier or any other parameter changes you better redo your qualification. To determine the quality of the weld best is to do a pre-production welding test and submit it to mechanical testing. To measure the HAZ you can ask a testcenter to investigate the microstructural material changes. If your process is stable you have some information about the length of the HAZ for FEM calculations.
For welding best rule of thump is place the joint at the area with the lowest strains as possible.
.
Are you an engineer and do you need to know more about a weld design for your application? Please type your question in the comment box below. If you are qualified I am willing to discuss your training needs or the design support you are looking for.
Other valuable input for engineers is also welcome and will be appreciated.
Aluminium greetings
Ellen. Many thanks for your input Kees Veeken!

door Ellen Vaders | apr 24, 2017 | alloy, corrosion, development
Aluminium is known for its corrosion resistance. Untreated aluminium applications like traffic signs and applications at sea are respected examples. Although aluminium is a relatively non noble material it protects itself via a passive oxide layer. Corrosion resistance of Aluminium is sensitive upon certain points in particular, these will be addressed in this blog.
To design aluminium without corrosion take the following aspects into account.
- pH value of the environment
- Nobility of Aluminium
- Crevice corrosion
- Composition of Aluminum
- Black corrosion
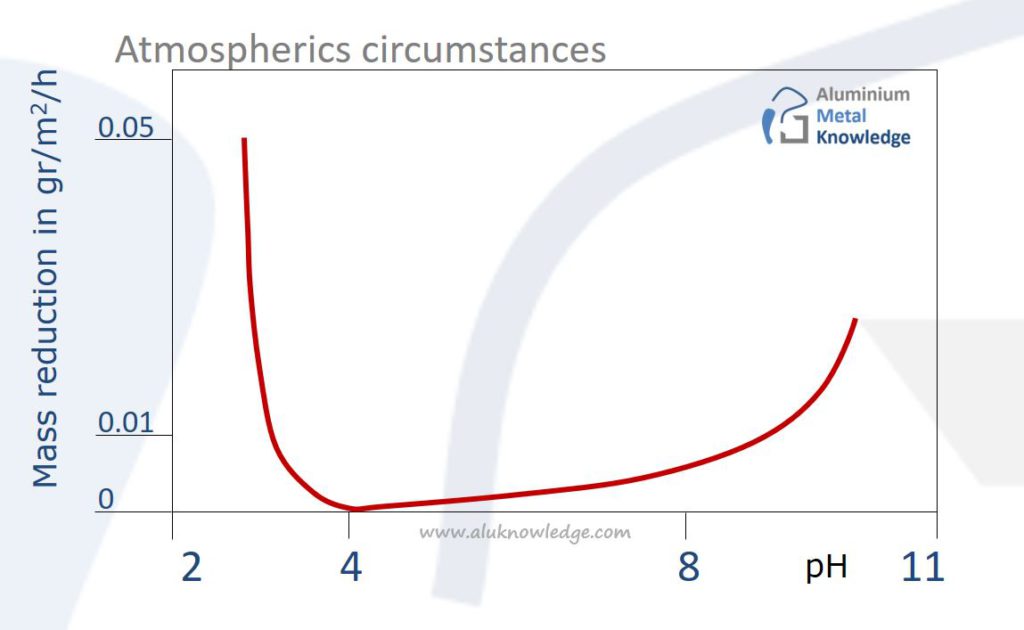
pH value of the environment
Corrosion resistance of Aluminium is influenced strongly by the pH value of the environment
For normal use, like cleaning, use fluids in combinations with Aluminium with a pH neutral value (7) to prevent corrosion.
To prepare Aluminium for surface treatment you can use this to principle to clean the surface with specific sour fluids. The surface needs to be neutralized and rinsed adequately afterwards to prevent corrosion issues!
Nobility of Aluminium
To avoid corrosion problems with Aluminium nobility of the material needs to be taken into account. Aluminium is a relatively non noble material and will therefore sacrifice itself on behave of more noble metals in moister environment. This leads to galvanic corrosion.
This is a serious aspect using Aluminium products, specialy in combination with other metal parts. To prevent this type of corrosion block out fluids, use the right combinations of metals in the same nobility level or isolate the metals from each other.

Crevice corrosion
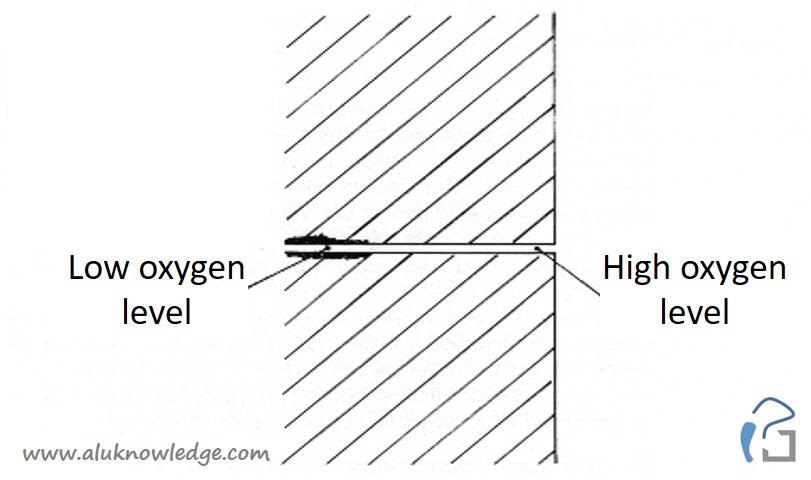
In a moister environment fluid will creep between narrow crevice due to capillary motion. To prevent crevice corrosion you have to avoid small crevices. Crevice corrosion occurs between crevices with a distance in between 0.2 mm to 0.5 mm. The solution for this matter is to increase crevice distance and reduce amount of contact surfaces. For example: a line contact is better than a surface contact.
With acid or salt fluids the potential for crevice corrosion increases.
Composition of Aluminum
Last but not least, the composition of the aluminum alloy influences corrosion resistance of Aluminium. If corrosion resistance is low you can surface threat this material in different ways. Take into account it needs more effort than with better corrosion resistance material.
If you take these mechanisms into account your design is robust against most common corrosion issues. In that case you take advantage of the natural corrosion resistance of Aluminium.
What was your biggest insight about corrosion issues so fare?
Let me know in the comment box below. This way, you’re helping me and others to design a world with optimal Aluminium use… Thanks in advance!

door Ellen Vaders | mrt 20, 2017 | alloy, aluminium, recycling
Technological advancement will increase. We need materials that are available in abundance and can be recycled with no, or as little as possible, impact to the environment. Aluminium recycling is endless.
Product disposal, waste?
When the lifetime of a product ends there are two options: put it somewhere as waste and get environmental pollution or reuse the materials. For each material the possibilities for recycling are different. The big advantage of Aluminium is that you can reuse it time and time again without loss of quality/capability.. Due to the low required energy for recycling and it’s “easy” process, Aluminium is wanted for recycling.
The value of 1 kg Aluminium is equal to 1 kilo recycled Aluminium. (as long as the alloy is the same of course, more about that later). This means Aluminium waste = money. So Aluminium waste equals money.
Still some people’s perception is, I need primary material, although this is not relevant for Aluminium. The recipe of the alloy is key for the characteristics, primary or secondary.
Each Aluminium product has on average already 52% recycled metal content in Europe. Since Aluminium is a relatively young material, industrial production was only possible after 1886 as opposed to e.g. the iron and it iron age around 1200 B.Chr.) we cannot supply the need at this moment with the current material available via recycling. (yet)
Alloy selection
The value of Aluminium scrap depends on the composition. Aluminium can be selected on form which determines composition.
Aluminium alloys can be selected separated at the manufacturer, uniform scrap has the higher perceived value by the recycler.
After its product life time you can easily make an alloy selection by separating on form. E.g. if a building is stripped place the profiles on the profile bin, plates together at the plates and knots in the casting corner. You have now a very good separation of the different alloys quite easily.
There are lots of ways to separate alloys some are very basic like above some are very sophisticated. Since Aluminium = money, it’s worth it.
The value of Aluminium scrap also depends on the purity. For standard commercial alloys, the more pure, e.g. 99,5 % Al, the cheaper the Aluminium; if you apply it for its application. This is because these low strength alloy’s needed minimum work to fabricate. On the other hand, for recycling, this type of Aluminium with a low level of impurities can be used in many different Aluminium recipes, so value of the scrap is relatively high.
Eddy current
For household waste several possibilities are used. About 70% of all the Aluminium throw away is captured and reused. A welcome advantage here is Aluminium = money. So instead of costing money to burn or landfill it delivers income. For separation different processes can be used. E.g. like eddy current.
Because Aluminium is a conducted light material, it is influenced by the rotating magnet with North-south poles. This generates an eddy current. Aluminium pieces crossing this area get accelerated and are separated.
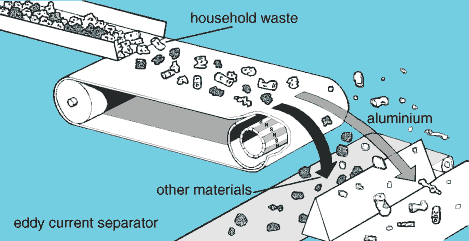
So if your product consists of different materials you can either design for disassembly, then separate on form or shred into parts.
For Aluminium the loop today is nearly closed. From all the Aluminium material ever produced 75% is still in use. For automotive and building the recycling percentage is above 95%. Since manufacturers know exactly which alloy they prefer, manufacturers offer more and more to take their product back in return at the end of its lifetime. Although the recycling system in Europe is quite efficient and for environmental reasons this is not necessary, economically it can be beneficial. The closed loop of Aluminium will become shorter and more efficient. Just by the fact Aluminium is worth it.
Whats your biggest question about aluminium recycling? Please put your answer in the commentbox below. It will be appreciated! Thanks in advance.

door Ellen Vaders | nov 28, 2016 | alloy, aluminium
Aluminium scandium alloys
With aluminium we can create all kind of forms. The material is ductile and form-able into all kinds of shapes. However an optimal design requires more effort to design. Start with your ideal form and then integrated functions.
What if we have something extra? Aluminium alloys which are 10 to 40% stronger? We will summarize the advantages on different areas when scandium is added in aluminium alloys .
First of all, strength of Aluminium alloys with scandium increases. This is shown if the graph above.
Next, recrystallization during coldwork (rolling) or extrusion is suppressed and prevented, which leads to better material quality.

Due to its fine grain refinement Scandium alloys reduces hot cracking in welds, increased strength in the welds and deliver better fatigue behavior. Welding filler/ thread with scandium has great potential for aluminium.

When will breakthrough appear?
Aluminium Scandium alloys are new and because of availability and price they aren’t popular jet. If they were used, it even happened that after product introduction the price of scandium was raised. On the other hand the amount of scandium used in aluminium was relatively high so price was effected drastically. Numbers of 10x material costs are mentioned.
This needs to be addressed before scandium will be commonly used in products.
Today, depending on the percentage of the scandium alloying element, the price of the aluminum with scandium will rice from 30% (at scandium levels of between 0.06 up to 0.12 wt% as used in Al-Zn-Mg (Cu) alloysand) up to e.g. 100%.
Price is under development. The process for mining and reduction is being optimized. Also a better quality scandium and lower quantities per alloy, generate a better proposition. For mining, new possibilities arise. This in total will be responsible for the acceptabele price levels. (make your comment if you want to know the source).

As mentioned in the past the scarcity leads to dramatic increase of price during application. And no one wants or can effort such surprises. Long term agreements are needed to solve this matter.
Main benefits scandium in aluminium alloys:
- Aluminium gets stronger
- Better weldability properties
- Grain structure during forming and extrusion stays intact
- Stronger at rising temperature, in combination with Zirconium.
Where do you want to use aluminium- scandium alloys in your products? Please write your comment below in the comment box and I sent you different sources for more information.
Would you like to stay informed about aluminium? schrijf je in, for English updates click here. Looking forward to your response!













