Aluminium is known for its corrosion resistance. Untreated aluminium applications like traffic signs and applications at sea are respected examples. Although aluminium is a relatively non noble material it protects itself via a passive oxide layer. Corrosion resistance of Aluminium is sensitive upon certain points in particular, these will be addressed in this blog.
To design aluminium without corrosion take the following aspects into account.
- pH value of the environment
- Nobility of Aluminium
- Crevice corrosion
- Composition of Aluminum
- Black corrosion
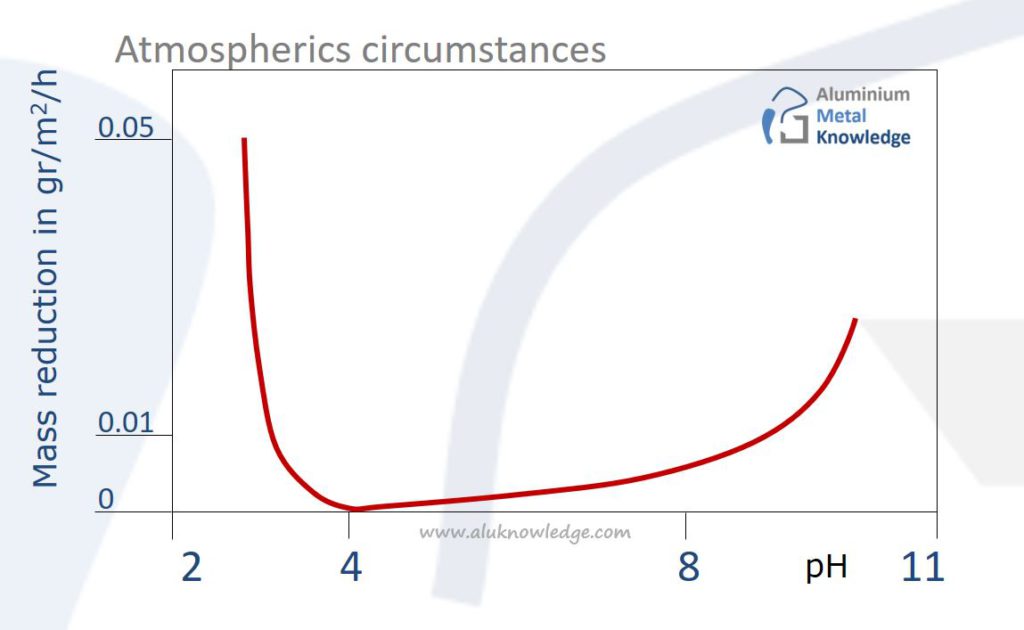
pH value of the environment
Corrosion resistance of Aluminium is influenced strongly by the pH value of the environment
For normal use, like cleaning, use fluids in combinations with Aluminium with a pH neutral value (7) to prevent corrosion.
To prepare Aluminium for surface treatment you can use this to principle to clean the surface with specific sour fluids. The surface needs to be neutralized and rinsed adequately afterwards to prevent corrosion issues!
Nobility of Aluminium
To avoid corrosion problems with Aluminium nobility of the material needs to be taken into account. Aluminium is a relatively non noble material and will therefore sacrifice itself on behave of more noble metals in moister environment. This leads to galvanic corrosion.
This is a serious aspect using Aluminium products, specialy in combination with other metal parts. To prevent this type of corrosion block out fluids, use the right combinations of metals in the same nobility level or isolate the metals from each other.

Crevice corrosion
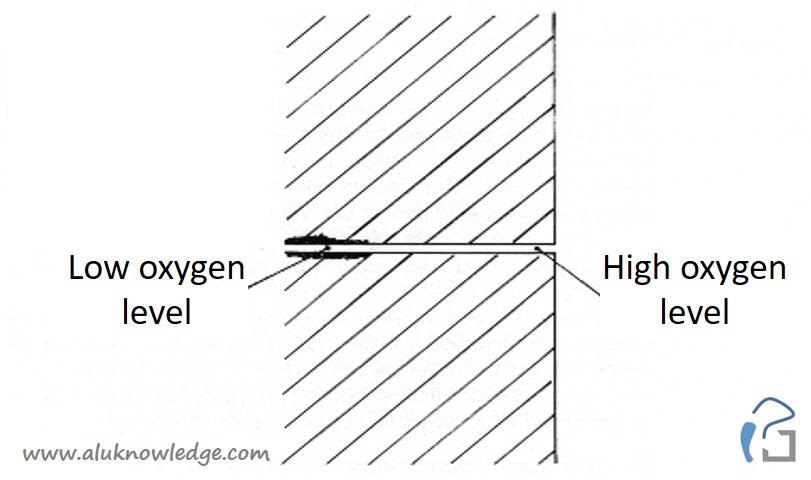
In a moister environment fluid will creep between narrow crevice due to capillary motion. To prevent crevice corrosion you have to avoid small crevices. Crevice corrosion occurs between crevices with a distance in between 0.2 mm to 0.5 mm. The solution for this matter is to increase crevice distance and reduce amount of contact surfaces. For example: a line contact is better than a surface contact.
With acid or salt fluids the potential for crevice corrosion increases.
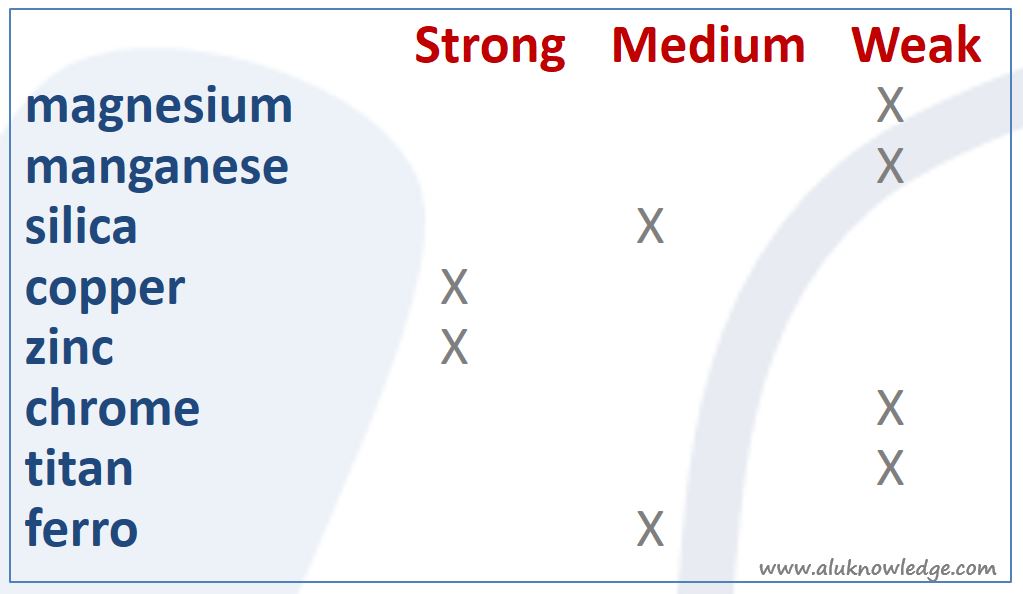
Composition of Aluminum
Last but not least, the composition of the aluminum alloy influences corrosion resistance of Aluminium. If corrosion resistance is low you can surface threat this material in different ways. Take into account it needs more effort than with better corrosion resistance material.
If you take these mechanisms into account your design is robust against most common corrosion issues. In that case you take advantage of the natural corrosion resistance of Aluminium.
What was your biggest insight about corrosion issues so fare?
Let me know in the comment box below. This way, you’re helping me and others to design a world with optimal Aluminium use… Thanks in advance!

Door hieronder je naam en e-mailadres in te vullen ontvang je direct de PDF over corrosie.


Hoi Ellen,
Leuk artikel misschien moet je ook even schrijven over de elektrische isolatie van Aluminium want dit kan ook meer corrosie veroorzaken.
Is bekend bij schepen en accu’s.
Groet Kasper
Leuke suggestie! thanks
Hoi Ellen,
Inderdaad een mooi artikel.
Als je echt iets bijzonders wilt maken met aluminium, relatief hoogbelast en in een corrosieve omgeving, moet je een aantal zaken terdege bekijken en overwegen.
Meestal zul je op basis van de berekening en de toepassing een bepaalde legering kiezen. Nu moet je aan de hand van veel beschikbare info controleren of de gekozen legering voldoet aan de corrosie-eisen.
Moet het werkstuk bijvoorbeeld geanodiseerd worden, dan is de vraag of de legering zich er voor leent.
Voor toepassing op zee zijn er bijvoorbeeld zeer geschikte legeringen voorhanden.
Gietlegeringen zijn bijvoorbeeld weer niet tot slecht te anodiseren. En ga zo maar door.
Kortom een corrosie-technisch verantwoord ontwerp is niet zo maar even snel gedaan.
Denk je dat dat de reden is dat men niet durft? of is dat door fouten in het verleden?
Als je de weg weet in de legeringen is het niet zo ingewikkeld, maar dan moet je de weg wel willen bewandelen of daar de juiste bronnen voor hebben. Ik ben met je eens dat er kennis van zaken benodigd is voor een goed corrosie ontwerp en dat geldt eigenlijk voor ale materialen.
Ellen,
Leuk samengevat stukje. Echter, je maakt niet duidelijk hoe men het tabelletje moet interpreteren.
Heeft bv Cu nu over het algemeen en sterk positief of negatief effect op de corrosiebestendigheid. Wij weten dat het een sterk negatief effect is bij Al. Maar bij staal kan Cu bv juist een sterk positief effect hebben. Dus ik denk dat het goed is hier wat helderder over te zijn.
Misschien nog eens wat aandacht voor aluminium toegepast als kathodische bescherming als alternatief voor Zn: minder milieu belastend, langere levensduur dan Zn mede dankzij het zelf-herstellend vermogen van Al.
Beste Erik,
Dank je wel voor je aanvulling!!